

Zinc Selenide ZnSe
ZnSe is used widely for IR components, windows and lenses, and for spectroscopic ATR prisms. Zinc Selenide is one of the materials of choice for CO2 laser optics operating at 10.6 microns.
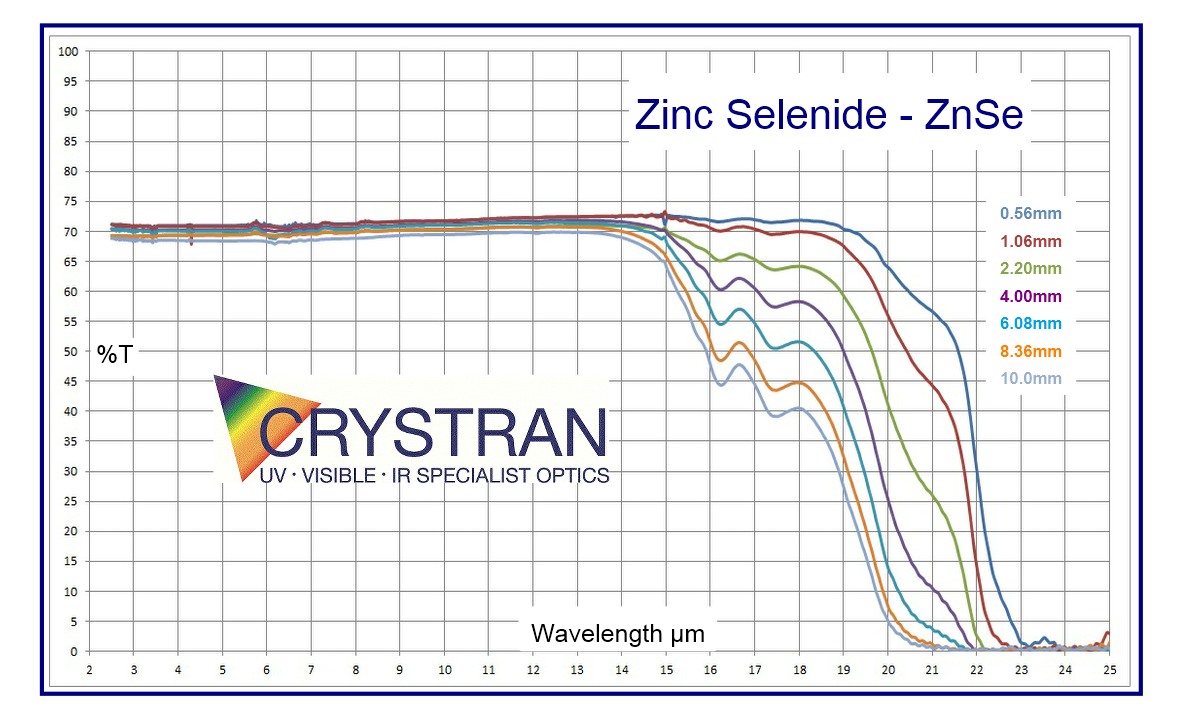
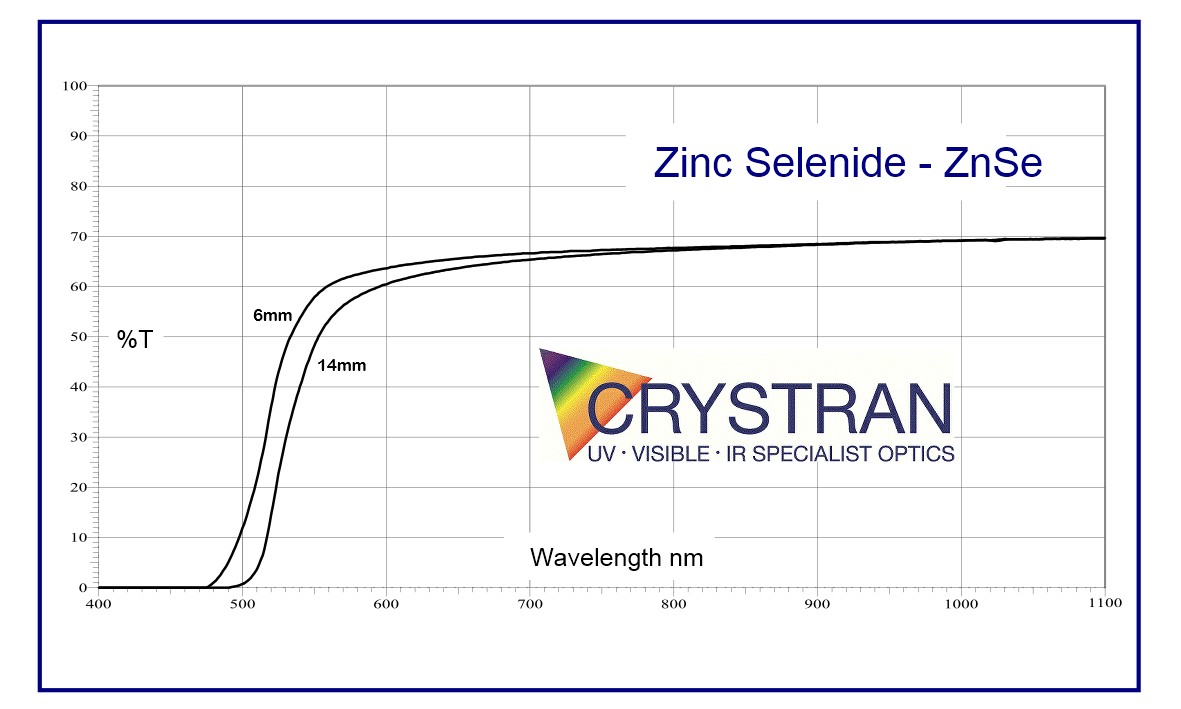
♦ ZnSe IR Transmission data ©2019. Open source with acknowledgement to Crystran Ltd.


Zinc Selenide oxidizes significantly at 300°C, exhibits plastic deformation at about 500°C and dissociates about 700°C. For safety, Zinc Selenide windows should not be used above 250°C in normal atmosphere
Download Zinc Selenide Guide to Cutting
Download Zinc Selenide Handling Guide
| µm | No |
|---|---|
| 0.54 | 2.6754 |
| 0.58 | 2.632 |
| 0.62 | 2.5994 |
| 0.66 | 2.5755 |
| 0.70 | 2.5568 |
| 0.74 | 2.5418 |
| 0.78 | 2.5295 |
| 0.82 | 2.5193 |
| 0.86 | 2.5107 |
| 0.90 | 2.5034 |
| 0.94 | 2.4971 |
| 0.98 | 2.4916 |
| 1.00 | 2.4892 |
| 1.40 | 2.4609 |
| 1.80 | 2.4496 |
| 2.20 | 2.4437 |
| 2.60 | 2.4401 |
| 3.00 | 2.4376 |
| 3.40 | 2.4356 |
| 3.80 | 2.4339 |
| µm | No |
|---|---|
| 4.20 | 2.4324 |
| 4.60 | 2.4309 |
| 5.00 | 2.4295 |
| 5.40 | 2.4281 |
| 5.80 | 2.4266 |
| 6.20 | 2.4251 |
| 6.60 | 2.4235 |
| 7.00 | 2.4218 |
| 7.40 | 2.4201 |
| 7.80 | 2.4183 |
| 8.20 | 2.4163 |
| 8.60 | 2.4143 |
| 9.00 | 2.4122 |
| 9.40 | 2.4100 |
| 9.80 | 2.4077 |
| 10.2 | 2.4053 |
| 10.6 | 2.4028 |
| 11.0 | 2.4001 |
| 11.4 | 2.3974 |
| 11.8 | 2.3945 |
| µm | No |
|---|---|
| 12.2 | 2.3915 |
| 12.6 | 2.3883 |
| 13.0 | 2.3850 |
| 13.4 | 2.3816 |
| 13.8 | 2.3781 |
| 14.2 | 2.3744 |
| 14.6 | 2.3705 |
| 15.0 | 2.3665 |
| 15.4 | 2.3623 |
| 15.8 | 2.3579 |
| 16.2 | 2.3534 |
| 16.6 | 2.3487 |
| 17.0 | 2.3438 |
| 17.4 | 2.3387 |
| 17.8 | 2.3333 |
| 18.2 | 2.3278 |
Zinc Selenide is produced by synthesis from Zinc vapour and H2Se gas, forming as sheets on Graphite susceptors. Zinc Selenide is microcrystalline in structure, the grain size being controlled to produce maximum strength. Single crystal ZnSe is available, but is not common but has been reported as having lower absorption and thus more effective for CO2 optics.
Zinc Selenide oxidizes significantly at 300°C, exhibits plastic deformation at about 500°C and dissociates about 700°C. For safety, Zinc Selenide windows should not be used above 250°C in normal atmosphere
REFERENCES
(1) Manufacturing Methods Program ZnSe Blanks. US Army R&D Feb 1980
(2) Manufacturers Published Data